News Story
BIOE Students Recognized for Innovative Healthcare Solutions at 2026 Capstone Design Expo
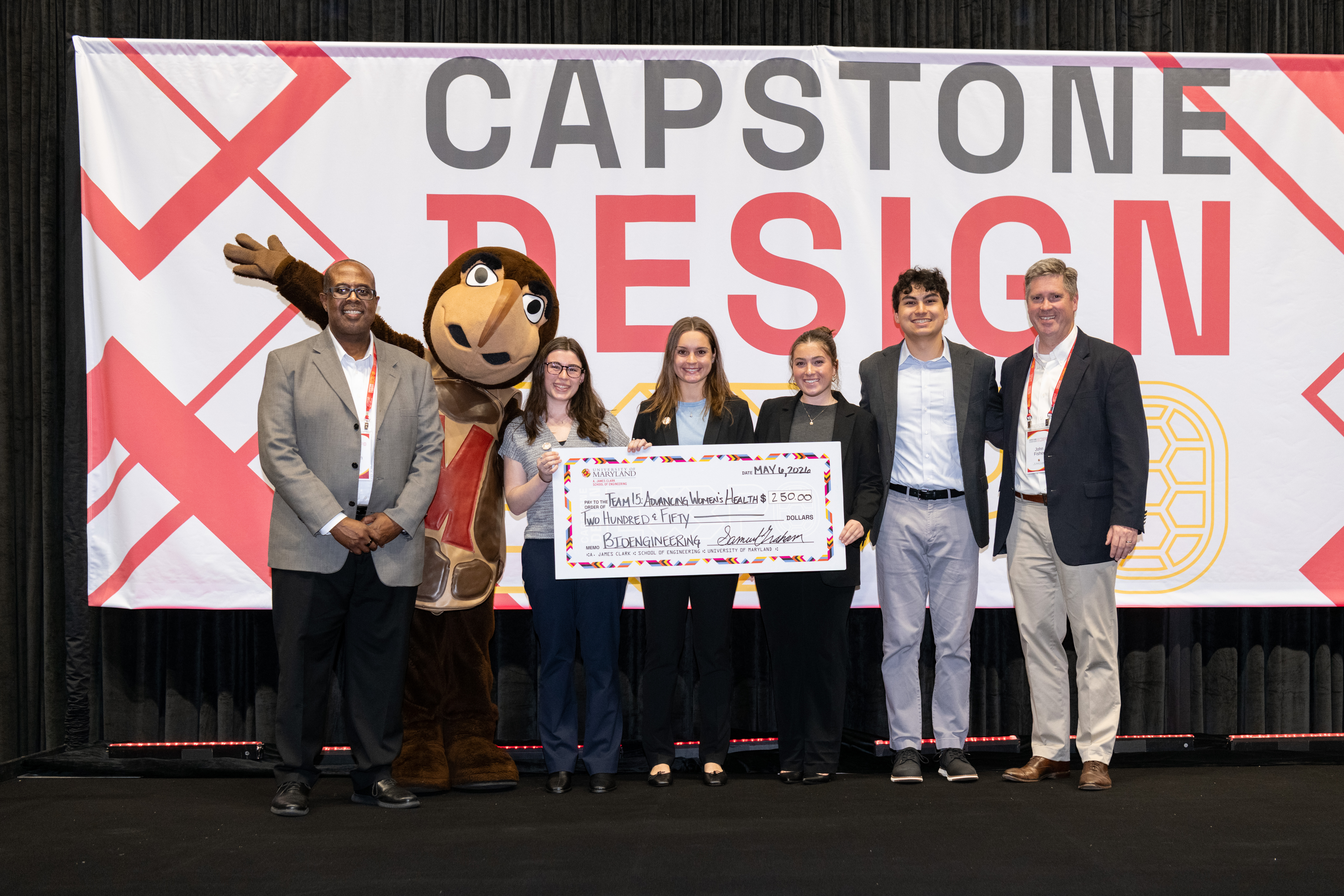
On May 6, the A. James Clark School of Engineering hosted its third annual Capstone Design Expo at the University of Maryland’s Xfinity Center, bringing together more than 760 engineering students from eight departments to showcase solutions to some of society’s most pressing challenges. The event featured 143 student projects and welcomed more than 1,000 visitors, including faculty, alumni, industry professionals, and community members.
Students from the Fischell Department of Bioengineering (BIOE) and Biocomputational Engineering (BCE) undergraduate programs presented projects focused on improving healthcare technologies, advancing patient safety, and expanding access to care. Several BIOE teams received top honors for their innovative and impactful designs.
BIOE Team 15 earned first place for their project, “Advancing Women’s Health Post-Surgical Care: Development of a Safe and Comfortable Vaginal Stent.” Current vaginal stents used after reconstructive surgery are often makeshift devices that can cause discomfort and hygiene concerns for pediatric and adolescent patients. The team developed a tear-drop shaped vaginal stent designed to improve stability, comfort, and healing outcomes while remaining low-cost and accessible. Their design demonstrated improved resistance to dislodgement compared to current standard-of-care devices and aims to improve post-surgical recovery for patients undergoing vaginal reconstruction procedures.
Second place was awarded to Team 6 for their “Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration (EBUS-TBNA) Training Model.” The team created an anatomically accurate and cost-effective simulation model to help physicians train for minimally invasive lung biopsy procedures commonly used to diagnose lung cancer and other diseases. By significantly reducing the cost of current training models, the project has the potential to improve physician training opportunities and patient outcomes.
Team 17 received third place for their “Proximity-Activated Patient Restraint System,” a device designed to help prevent ICU patients from unintentionally removing critical medical devices such as endotracheal tubes and IV lines. Their inflatable glove system activates only when a patient’s hand approaches sensitive equipment, preserving patient mobility while reducing the risk of self-extubation and associated complications.
The Award for Translational Design, sponsored by the Fischell Department of Bioengineering Advisory Board, was received by Team 7 for their project “High-Flux Hemofiltration System for Toxin and Solute Removal.” The award recognizes the team with the highest overall design score and honors projects that demonstrate strong problem identification, thoughtful engineering design, and translational potential. Team 7 developed a high-flow hemofiltration system that combines extracorporeal membrane oxygenation (ECMO) and dialysis technologies to improve toxin removal in critically ill patients while reducing resource use and clinician workload.
Team 11 received the MPower Award for their project “Automated Capillary Refill Time Monitor for Peripheral Blood Flow Assessment in Shock Patients.” Sponsored by the University of Maryland MPowering the State Strategic Partnership, the award recognizes exemplary collaboration between the University of Maryland, College Park and the University of Maryland, Baltimore. The team developed a portable device that automates capillary refill time measurements to improve the monitoring and treatment of septic shock patients while reducing clinician variability and bias.
This year’s projects addressed a wide range of healthcare challenges, including hemorrhage control, respiratory care, neurological disease monitoring, medical imaging, women’s health, and physician training. Through hands-on collaboration with clinicians, researchers, and industry mentors, students applied engineering principles to develop solutions with real-world clinical impact.
The Fischell Department of Bioengineering extends its gratitude to this year’s Capstone judges, including members of the BIOE Advisory Board, faculty, clinicians, and industry professionals who supported the competition and provided valuable feedback to student teams.
BIOE’s Capstone Design competition is supported by the Fischell Family, the University of Maryland MPowering the State Strategic Partnership, and Expo event sponsor Whiting-Turner (WT). In addition, BIOE Capstone students benefit from mentorship provided by clinicians and industry professionals, including collaborators from the UMD School of Medicine, Children’s National Health System, AstraZeneca, Air Force Research Laboratory, and the Robert E. Fischell Institute for Biomedical Devices.
Team 1: The Mucus Matrix
Ana Kanazir, Jonathan Killoran, Neelay Sachdeva, Supriya Shastry, Catarina Teixeira Fernandes
Advisors: Dr. Yang Tao / Sponsor: Dr. Jeffrey D. Hasday
In the intensive care unit (ICU) and other hospitalized respiratory settings, sputum characteristics serve as vital indicators of airway infection, inflammation, and mucociliary clearance efficiency. Despite its diagnostic value, current assessment relies heavily on subjective visual inspections, leading to high inter-clinician variability and potentially delayed therapeutic interventions. This project addresses this gap by developing a Point-of-Care Quantitative Sputum Analyzer (PQSA), a compact bedside device designed to provide objective, numerical assessments of key sputum properties within 10 minutes using a minimal 3-10 mL sample. The PQSA utilizes an absorbent polyethersulfone (PES) membrane to isolate the clinically relevant jelly fraction for dual-mode analysis: (1) an optical module that employs Hue-Saturation-Intensity (HSI) algorithms to quantify myeloperoxidase (MPO)-related inflammation, and (2) an elasticity module that measures structural deformation in response to a controlled air puff using a laser-camera system. Major prototyping results have successfully validated a Raspberry Pi-based image-processing pipeline capable of isolating laser-line displacement on artificial sputum substitutes, translating these physical changes into a standardized Bronko Test color score and an effective elastic modulus. To ensure reproducibility, we finalized a 3D-printed CAD housing that integrates ring LED lighting for consistent illumination and a custom syringe pump for precise, repeatable air-puff delivery. Beyond improving diagnostic accuracy, the PQSA addresses ethical impacts by standardizing care to reduce clinician bias and prioritizing global equity for high-risk respiratory regions. Furthermore, the design promotes environmental sustainability by utilizing a disposable internal sample holder to minimize medical waste while maintaining biosafety standards.
Team 2: Improving CPR to save lives
Ethan Bender, Eleni Green, Christine Needham, Heather Neisser, Pantea Vafaei
Advisor: Beatrice Kondo / Sponsor: Michael Shoykhet
Cardiac arrest is one of the most deadly medical emergencies, reported as the third most common cause of death in the United States. Many treatments have risen to address this problem. Through all, cardiopulmonary resuscitation (CPR) has proven to be the cornerstone of cardiac arrest treatment, however CPR quality can vary depending on many factors, including clinician training and the patient’s age. This disparity in treatment can be the difference between life and death. There is evidence to suggest that CPR quality may be improved through real-time feedback, due to increased levels of diastolic blood pressure of patients undergoing effective CPR compressions. However, there is no device capable of providing physiologically relevant data in the field. Current methods are either too invasive or too slow for actionable feedback outside of the hospital setting. Our goal is to address this need. We are building a monitor that leverages unique material properties and force distributions to collect physiological data on cardiac arrest patients’ condition, providing rapid, patient-specific CPR feedback and helping first responders save lives.
Team 3: GreenCell Reprocessing
Kelsey Afoakwa, Jay Gonski, Mabel Heine, Ighelose Ohonsi, Jared Ross, Surabhi Singh
Advisors: Dr. William Bentley, Dr. Edward Eisenstein / Sponsor: Mr. Philip Ceary, Astrazeneca
Large-scale antibody production at Astrazeneca (AZ) generates a significant amount of cell media waste, which has high waste management costs as well as contributes to negative environmental impact due to carbon emissions. The goal of this project is to find a secondary sustainable use for the waste media. This project proposes a process that uses microalgae to decrease the carbon content in spent media to decrease the emissions into the environment and generate valuable biomass as a byproduct. Microalgae has proven cleaning properties that show the uptake of carbon, nitrogenous waste, phosphorus, etc. from the media. The biomass created as a byproduct of the process has strong potential as supplemental feed for oyster hatcheries as it is known to have sufficient nutritional properties to sustain oysters’ entire growth cycle. Saltwater microalgae were grown in spent cell media, and their growth rates and carbon uptake were assessed. We developed a material balance model using MATLAB that predicted the produced biomass, amount of CO2 captured per day, and the biological oxygen demand changes. A proof-of-concept experiment was conducted to show successful growth of six saltwater species in spent cell media. Preliminary results show effective growth of the saltwater microalgae species through absorbance readings and cell counts. With our proof-of-concept data and material balance predictions, our project is proven to be scalable and sustainable as a means of decreasing the carbon footprint of AZ’s antibody production and other biopharmaceutical processes.
Team 4: Titer Titans
Praneet Boggarapu, Chloe Connolly, Sarah McHugh, Prishasai Ramnath, Cameron White
Advisors: Dr. Allan Doctor and Dr. Deborah Goldberg / Sponsor: University of Maryland School of Medicine
Massive hemorrhage is responsible for nearly 90% of preventable trauma-related deaths, and mounting evidence supports the use of early whole blood (WB) transfusion over packed red blood cells alone to address both coagulopathy and shock. Group O-negative WB is preferred in these urgent scenarios where ABO typing is not practical. However, its plasma contains anti-A and anti-B isoagglutinins that can cause severe hemolytic reactions in non-O recipients. The current standard of practice to address this issue relies on “low-titer” Group O-negative WB (LTOWB), which has limited donor availability, especially in urgent trauma scenarios. To overcome this barrier, we propose an automated antibody removal system utilizing the Therasorb Omni immunoadsorption column from Miltenyi Biotech, a CE-marked technology that removes immunoglobulins via sepharose-bound polyclonal anti-human Ig antibodies. By integrating this column into a streamlined blood-bank workflow featuring pumps, solenoid valves, conductivity sensing, peristaltic pumping, and a sterilizable casing, we aim to selectively eliminate anti-A and anti-B antibodies from Type O-negative plasma. This device will preserve the efficacy, safety, and integrity of WB for hemostasis, oxygen delivery, and volume expansion to improve patient outcomes in emergency trauma situations. Successful implementation of this system would significantly expand the donor pool for universal whole blood transfusion and enhance success in massive transfusion protocols for trauma care.
Team 5: Blood Filtration for Autologous Transfusion
Edward Bang, Dana Harel, Jackson Laub, Rebecca Lu, Jessica Mense, Stephen Maxwell Wiafe-Amoako
Advisor: Dr. Steven Jay / Sponsor: Dr. Elizabeth Powell and Piersson Hoenisch of ARC3 Laboratories
While hemorrhaging remains one of the leading causes of death from traumatic injury, it is still quite limited in terms of available interventions. Currently, the most widespread and standard of care treatment for hemorrhagic injuries is by way of allogenic blood transfusions, a limited resource that gets continually bottlenecked by supply chain, storage, and accessibility. This project aims to design and develop a compact blood filter able to sanitize blood for safe autologous retransfusion. Using affinity-based filtration with polymyxin B immobilized on a mesh, our filter will selectively remove bacteria and endotoxins while preserving red blood cells. Through iterative prototyping, we evaluated multiple tube geometries and mesh densities using benchtop flow experiments. The final geometry chosen was a tube of 8 in length and 1 in inner diameter with a mesh surface area of 4 ft2. The flow rate and final geometries were evaluated in a fluid dynamics model with a final flow rate of 49.6 mL/min, 3 minute residence time, and shear stress of 0.0045 Pa. Testing of bacterial binding to an aqueous solution of polymyxin B revealed that a 10^6 times greater concentration of molecules to CFU is necessary for complete binding. Additionally, a residence time of at least 5 minutes is required for the system to plateau near maximal bacterial killing. Future testing using blood will be necessary to confirm blood clotting behavior. This device promotes equitable access to care by providing a low-cost, portable solution that can be used in resource-limited or emergency environments where donor blood is unavailable. Overall, this system has the potential to improve survival outcomes by enabling rapid, on-site blood filtration and reuse in critical situations.
Team 6: EBUS-TBNA Training Model
Yassmin Abbouchi, Raifah Alam, Emmett Bachran, Mia Jocic, Eric Lee
Advisors: Dr. John P. Fisher / Sponsor: Dr. Gaurav Ajmani, Pulmonary Critical Care, University of Maryland School of Medicine
SECOND PLACE
Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration (EBUS-TBNA) is an essential procedure in diagnosing and staging lung cancer, sarcoidosis, and lymphoma. EBUS-TBNA enables minimally invasive sampling of mediastinal and hilar lymph nodes. This procedure utilizes a convex, side-viewing endobronchial ultrasound scope in combination with a biopsy needle to endoscopically sample lymph nodes and masses. Together, they allow for real-time visualization of anatomical landmarks during transbronchial needle aspiration. Despite the widespread clinical use of EBUS-TBNA, there are limited models available to accelerate procedural proficiency that are both anatomically accurate and cost-effective. EBUS-TBNA trainees are often limited to clinical encounters (live patient procedures) and require the guidance of an experienced clinician. Existing models with high fidelity are cost-prohibitive and are sold at the scale of thousands of dollars. To address these limitations, we created a $115 model that incorporates second-generation bronchi, 10 commonly biopsied lymph node stations, and blood vessels frequently used as anatomical EBUS landmarks. Through development and integration in clinical settings, this model will provide realistic needle feel and echogenicity during EBUS-TBNA simulation to streamline the training process, enhance procedural proficiency, and improve patient outcomes.
Team 7: High Flux, High Flow Hemofiltration
Gage Griffith, Rohan Gudwani, Grace Herron, Nicholas Kalbaugh, Emilee Yuan
Advisors: Dr. Allison Grazioli, Dr. Joshua King, Dr. Silvina Matysiak / Sponsor: University of Maryland, School of Medicine
Translational Award
Acute conditions such as sepsis, crush injuries, and poisonings can lead to the accumulation of harmful, non-dialyzable toxins in the bloodstream. Current clinical practice addresses this limitation by using hemodialysis in tandem with Extracorporeal Membrane Oxygenation (ECMO), enabling simultaneous life support and enhanced toxin transport. However, this approach is constrained by a significant mismatch in flow rates, as ECMO operates at 4-5 L/min, while dialysis systems function at 0.1-0.5 L/min. To address this critical gap, we developed a high-flow hemofiltration system that integrates ECMO and dialysis into a single platform, enabling improved toxin clearance while maintaining hemodynamic stability. Our prototype introduces a secondary recirculation loop that processes effluent from the hemodialysis filter through a custom adsorption column. This column contains an ion-exchange resin designed to selectively bind toxins, enabling their removal from the effluent, while essential electrolytes are preserved and returned to the bloodstream, maintaining physiological balance. By regenerating and reusing the filtrate, this design eliminates the need for continuous replacement fluid, significantly reducing manual intervention, resource consumption, and waste generation. Our prototype uses potassium as a hyperkalemic model toxin. The system incorporates glass beads and glass wool to stabilize inlet and outlet flow and ensure uniform distribution. A sodium and calcium polystyrene based ion exchange resin is used to selectively bind potassium ions, while a fine membrane barrier prevents resin particle migration into the effluent stream. The adsorption column is centrally positioned within the system and sealed using a water-resistant sealant to maintain structural integrity and prevent leakage. Ethically, equitable access across diverse healthcare settings must be prioritized, particularly for rural and low-resource hospitals. In early clinical use, clear informed consent is essential due to the novel nature of the system, and use in non-consenting patients should be deferred until sufficient validation is achieved. Rigorous testing and adherence to regulatory standards are necessary to ensure safe and responsible clinical implementation. However, this device has the potential to improve patient outcomes and reduce clinical workload while increasing hospital efficiency.
Team 9: Blood Recollection from Traumatic Injury
Vicky Chen, Noah Hill, Alexis Johnson, Adedamola Owolabi, Carly Peterson
Advisor: Dr. J. Helim Aranda-Espinoza, Dr. Nan Xu / Sponsor: University of Maryland, School of Medicine
Hemorrhage remains the leading cause of preventable death in combat and prehospital trauma, particularly for abdominal and junctional injuries where tourniquet and direct pressure are ineffective (Combat Casualty Care Research Program, 2023). In combat settings, limited access to donor blood, lack of cold storage, and prolonged evacuation times significantly increase mortality risk during the Golden Hour. This project proposes the design and development of a combat-ready blood recollection system that allows the safe collection and storage of a patient's own blood for potential autologous transfusion in resource-limited environments. The proposed device is designed to be placed directly over an open wound, where it gently collects shed blood while minimizing additional tissue damage and blood cell hemolysis. The system integrates three core elements. First, a biocompatible porous sponge made of polyurethane foam absorbs pooled blood from irregular wound shapes. Second, a sealed adhesive interface maintains an airtight environment and prevents air entry. Third, controlled negative pressure generated by a rotor pump draws blood into a small anticoagulant-compatible reservoir. Together, these components enable efficient blood recollection while preserving blood integrity for potential reinfusion. The system specifically targets abdominal and junctional hemorrhage, injuries that are difficult to manage with existing technologies and account for a large proportion of preventable combat deaths. Unlike traditional cell salvage systems that require sterile surgical environments and bulky equipment, this device is compact, portable, and designed for use by minimally trained providers in high-stress field conditions. The design prioritizes simplicity, rapid deployment, single-user operation, and single-use sterile patient-contacting components. Overall, this approach aims to reduce dependence on donor blood and improve survivability during prolonged casualty care.
Team 10: MigraSense
Tinebte Daniel, Althea Eclarin, Ryan Jerome, Simone Robinson, Abigail Serata
Advisor: Rohan Fernandes / Sponsor: Raquel Langdon
This project focused on building an electrochemical biosensor to measure levels of calcitonin gene-related peptide (CGRP) in the blood to help monitor headaches for migraine patients. The sensor uses a specially coated electrode that can recognize and bind to CGRP. When CGRP attaches, a redox reaction occurs that changes the flow of electrons at the electrode, creating an electrical signal. The strength of this signal is related to how much CGRP is present, and it can be measured using simple electrochemical methods. Because CGRP levels tend to increase during migraine attacks, this sensor makes it possible to track changes over time. This information can then be used to better understand a patient’s migraine patterns and adjust treatments, helping create more personalized and effective medicine plans. It also benefits patients directly by helping them better predict when headaches may occur, allowing them to take action earlier and reduce the overall burden of the disease.
Team 11: P.R.E.S.S. (Perfusion Refill Evaluation for Septic Shock)
Renee Jaranson, Roshni Pallavajjala, Scott Regan, Riley Schoch, Kevin Zhang
Advisor: Dr. Ian White / Sponsor: Dr. Mohammad Abdulelah
MPOWER AWARD
Septic shock, the most severe form of sepsis, results from an overactive immune response to infection and is the current leading cause of death in US hospitals. Capillary refill time (CRT) acts as a clinical metric used to quickly assess peripheral blood flow, as drops in peripheral perfusion occur in extremities during septic shock. It has been shown that the use of personalized CRT measurements results in improved clinical outcomes in the treatment of shock patients. However, CRT measurements are incredibly subjective and inaccurate due to variability in environmental conditions and clinician to clinician measurement. This project addresses these deficiencies in developing a device capable of taking automated CRT measurements while also accounting for sources of variability. The successful implementation of this device would provide a comparably cheaper, and reliable solution for monitoring patient condition than the current standard of care, serum lactate testing. We have successfully developed a prototype with portable housing that recapitulates all key steps in taking CRT measurements, including a working circuit and accompanying software that provide adequate blanching force, continuous sensing of finger perfusion, and an OLED screen to display CRT measurements. The prototype is also designed to be invariant to common sources of clinical bias, namely skin type and skin tone. Through further development and deployment in clinical settings, this device aims to alleviate sources of bias through standardizing care, and improve shock patient outcomes with rapid point-of-care monitoring.
Team 12: The Wound Washers
Kaviya Kandaswamy, Zoya Naseem, Alina Sharma, Wilhelm Smith, Jane Zhang
Advisor: Dr. Matthew Grzywinski and Dr. Catherine Kuo / Sponsor:?
As of 2023, more than 12 million patients are seen each year in emergency departments (EDs) for wounds and lacerations. The initial step of wound care is irrigation, which removes foreign materials and reduces infection risk. However, wound irrigation is currently a manual process: clinicians must repeatedly irrigate with syringes or punctured saline bottles, which is physically strenuous, time-consuming, and often messy due to saline runoff. Furthermore, the process is subject to user variability, as fluid pressure and volume of saline delivered depends on the clinician. Our poster demonstrates a novel device that provides a hands-free solution to puncture wound irrigation (Figure 1). We demonstrated considerable progress towards standardizing the volume (250mL of fluid per 5 cm of wound dept) and pressure (15-18 psi) of saline delivered. Our device also contains functional features such as an irrigation shield to prevent splashback, all under $25 per unit. We conducted benchtop testing comparing our device to the standard of care (syringe) using a silicone wound packing model to demonstrate debris removal. With this device, ED clinician workload is reduced, infection risks are lowered, and overall quality of care in the ED is improved.
Team 13: VitalPatch
Abtin Alizadeh, Aiden Bellamy, Alie Bogusch, Jack McCann, Jai Vaichalkar
Advisor: Dr. Sara Molinari / Sponsor: Dr. Nico Dimenstein, Children's National Hospital
The VitalPatch is a point-of-care medical device focused on monitoring vitals, including pulse presence and limb temperature, in pediatric emergency medicine settings. Dr. Nico Dimenstein from Children's National Hospital expressed his negative experiences with manual pulse checks, including wasted man-power, time constraints, and some errors in standard vital detection methods utilized across emergency response teams and pediatric intensive care units. The VitalPatch offers a solution by actively monitoring the patients extremities for the entirety of the time the device is placed on the wrist or foot, eliminating the need for manual pulse checks. The device consists of a small, 3D printed casing with temperature and pulse sensors, making it a cost effective alternative for ambulances or emergency responses. This is a single use device that utilizes a customized circuit, including the sensors, visual cues, and an organic light emitting diode (OLED) screen, as well as continuous glucose monitoring (CGM) tape that is able to be safely stuck to the extremity of interest for as long as needed. Prototyping results have been promising, as Team 13 have achieved consistently accurate IF/NOT gated feedback for the pulse that allows for presentation on the OLED screen, as well as a temperature sensing mechanism that consistently measures temperature and variance over 30 minute periods. The temperature sensor has been validated against multiple hospital gold standards, including infrared-based thermometers, demonstrating an accuracy within +/- 1 °C of clinically relevant body temperature measurements, and testing by taking an accurate room temperature, placing on the wrist, and achieving clinically relevant results in the same trial. The pulse sensor has also been validated against the gold standard pulse oximeter in which the device has demonstrated an accuracy within +/- 5 bpm. Key ethical considerations for the VitalPatch include accuracy and reliability, patient safety, and accessibility. False positives or negatives in critical care settings can lead to serious clinical consequences, they must be minimized through rigorous design and validation. The device's limitations must be clearly communicated, as the VitalPatch is a monitoring aid and not a diagnostic tool. Patient safety is ensured through the use of non-allergenic materials and safe battery operation to prevent harm during prolonged skin contact. Finally, the device has been intentionally designed to be low-cost and aligned with point-of-care standards, ensuring broad accessibility across diverse clinical environments.
Team 14: BDNF Measurement Device
Georgia Hammond, Anika Hukmani, Talia Levine, Nuha Talukder
Advisor: Dr. Alexander Xu, Dr. Rachel Reoli
Brain-Derived Neurotrophic Factor (BDNF) is a protein biomarker linked to overall neurological and health and cognitive function. Depleted levels of BDNF are associated with neurological disorders such as Alzheimer's, Parkinson's disease, and depression; therefore monitoring BDNF levels can help track disease progression and guide individualized rehabilitation and physical therapy treatment. The current measurement standard is a blood-based ELISA test, which requires invasive blood draws, lengthy processing times, and specialized laboratory equipment. This process is uncomfortable for patients and impractical for frequent biomarker quantification. There is a need for non-invasive sample collection and an improved monitoring process with decreased diagnostic time. To address this need, our team tested the feasibility of a saliva-based ELISA to improve patient experience and developed a portable plate reader to decrease protein quantification time. We learned that BDNF is not present in detectable levels in saliva using our ELISA kit which is valuable information for future work. We calibrated a portable plate reader to find concentration from absorbance level for our ELISA standard curve. The ethical impact of our device is overall positive for patients and physicians as it is less invasive than blood sampling, increases sampling frequency, and enables BDNF to be used as a biomarker for treatment regiments.
Team 15: Novel Vaginal Stent
Lena Chennat, Haley Decker, Leah Falk, Selma Hamza, Anthony Liberatore
Advisor: Dr. Erika Moore / Sponsor: Children's National Hospital
FIRST PLACE
Vaginal reconstructive surgery in pediatric and adolescent patients is a relatively rare but critically important procedure, typically performed for congenital anomalies. In the surgery, a vaginal canal is created or reconstructed using native tissues or grafts. Vaginal stents are used to maintain the patency and shape of the canal during the post-operative healing period, which can last several weeks to several months. Current practice often relies on makeshift, condom-based stents, which present challenges in hygiene, stability, and patient comfort. To address these limitations, this project developed a novel, tear-drop shaped vaginal stent with a compression feature, engineered using a 16 French catheter layered with Coban wrap, pre-wrap foam, hydrocolloid, and a parafilm outer seal to balance structural support, flexibility, and biocompatibility. Mechanical testing demonstrated an average retention force of 7.85 N ± 0.98 N, indicating improved resistance to dislodgement compared to control stents simulating the current standard of care. Clinically, our new stent design improves stability and the material design has the potential to reduce complications such as pain, malodor, and worsening vaginal stenosis, thereby improving patient comfort and healing outcomes. At the same time, the use of vaginal stents in pediatric populations raises important ethical considerations, as patients and their families may experience significant emotional distress alongside physical burden, impacting overall quality of life. Overall, this women’s health project aims to provide a low-cost solution to a complex problem that will improve awareness and accessibility for these vital procedures.
Team 16: Built to Breathe
Azkah Anjum, Savannah Phillips, Shrish Pokharel, Sanjana Vandanapu, Athena Yang
Advisor: Dr. Zhongjun Jon Wu, Dr. Tao Lowe, Dr. Li-Quan Zhang
In cases of severe respiratory distress, venovenous extracorporeal membrane oxygenation (VV-ECMO) is a life-saving therapy that supports patients by performing the function of the lungs. VV-ECMO handles delivery of oxygen to the blood as well as removal of carbon dioxide by adjusting the blood flow and sweep gas flow. Current VV-ECMO devices require manual adjustment of these parameters, which involves constant monitoring from trained professionals, including perfusionists and nurses. Immediate treatment is difficult to achieve with the burden of manually adjusting VV-ECMO settings, especially in low-resource environments. To eliminate the need for direct manipulation of VV-ECMO, we developed an autonomously regulated VV-ECMO system to facilitate oxygenation and carbon dioxide removal in critically ill patients. Our design utilizes a sensor-driven closed-loop control framework, combining fuzzy logic with proportional-integral derivative control to manage system dynamics. During each cycle, the system collects key measurements, interprets them on a continuous spectrum, and issues gradual adjustments to sweep gas flow and blood pump speed. While our benchtop prototype demonstrates the feasibility of this approach, we must acknowledge that our simulated conditions may not perfectly replicate the complexities of a human patient, thus the system requires further testing before it can be installed in hospitals. Automation of ECMO will expand equitable access to advanced respiratory support by reducing the specialized labor required, allowing more hospitals to offer ECMO safely while improving patient safety.
Team 17: Proximity-Activated Patient Restraint System
Seth Boledovic, Lauren McLaughlin, Tyler Moran, Nyan Shipul Patel, Alena Zheng
Advisor: Dr. Jeffrey Hasday (Clinical Mentor), Dr. Giuliano Scarcelli (Faculty Mentor)
THIRD PLACE
The intensive care unit (ICU) has various critical devices, such as endotracheal tubes (ETTs) and intravenous (IV) lines, that support patient recovery. Yet, patients can unintentionally or purposefully detach these devices. This phenomenon, known as self-extubation, can lead to further medical issues and re-intubation procedures, extending the patient’s stay in the ICU and adding up to $25,000 in costs. Current prevention strategies rely on either physical restraints, heavy sedation, and/or mechanical securement devices. However, these strategies are also associated with serious patient complications, including delirium, PTSD, and patient discomfort. Therefore, there is a clear unmet clinical need for a safe, effective solution that prevents self-extubation while preserving patient comfort, dignity, and mobility. Our solution is a custom glove that houses an inflatable bladder, connected to a solenoid-valve-based inflation and deflation system. Coupled with a two-camera system developed by a past Capstone team that tracks patient hand position relative to key devices, our device activates and triggers inflation when a patient’s hand approaches a critical device, impeding the patient’s IP joints and preventing hand closure. When the patient’s hand is not detected near key devices, the glove deflates to allow the patient to regain full mobility. Our device provides full patient autonomy when inactive and prevents patients from grasping these key devices with instantaneous activation. Patients will benefit from increased mobility in the ICU, thereby improving patient outcomes and reducing discomfort. Our device also gives healthcare providers more time to intervene, removing stress and decreasing the overall cost of complications for hospital systems.
Team 18: Microfabricated Scaffold for Epilepsy Therapy
Faith Harden, Kalina Li, Shreeji Patel, Caroline Rosenberry, Elise Smith
Advisor: Dr. Jeffrey Hasday (Clinical Mentor), Dr. Giuliano Scarcelli (Faculty Mentor)
Epilepsy is the 4th most common neurological disorder that causes seizures in humans (Epilepsy Foundation). The basis of epilepsy is not fully understood, so current treatments can be ineffective or only provide symptomatic relief. Long-term use of these treatments can have severe side effects and present an economic burden to patients. Alternatively, our project aims to optimize in vivo epilepsy modeling of brain organoids derived from pluripotent stem cells (PSCs), which will model gene expression of cortical cells. Due to diffusion challenges typically presented by 3-D cell culture, a porous scaffold was designed to support the growth and maturation of PSCs into brain organoids, incorporating an inner media reservoir to prevent necrotic core development. The prototype is a 1425 μm radius spherical scaffold with two 450 μm concentric layers for cell adhesion. For seeding purposes, there are 300 μm diameter pores on each layer. The prototype was transferred to Children’s National Hospital for experimental testing of PSC culture, evaluating cell viability, self-organization, and functionality. Ultimately, we hope to develop an improved neural model of epilepsy to offer a promising alternative to conventional medications.
Team 19: Smart Tourniquet
Daniel Erickson, Haider Hussain, Michael Kim, Sean Leonard, Rafael Joseph Martinez Virador
Advisor: Dr. Brian Blair / Sponsor: Airforce Research Laboratory
This project proposes the design of a compact, lightweight, and easy-to-use “smart tourniquet” that preserves the proven effectiveness of conventional windlass tourniquets while integrating additional features to mitigate time-dependent complications. Key proposed elements include an automatic, fail-safe timing mechanism to track the duration of application, intuitive indicators to alert medics when safe time thresholds are exceeded, and a design that facilitates safe reassessment or controlled release without compromising hemorrhage control. The ultimate goal is to reduce preventable limb damage and systemic injury while maintaining simplicity, reliability, and compatibility with existing military training and battlefield conditions.
Team 20: The Tetramers
Arinze Ezeifeka, Blake Gilbert, Gabriel Lipman, Janet Mwebi
Advisor: Dr. Lan Ma / Sponsor: Dr. Valentin Stanev, AstraZeneca
Antibody development is central to modern therapeutics, but generating experimental data is prohibitively expensive; even measuring around 10,000 sequences can cost between $1.5M and $8M. Machine learning offers a way to predict developability properties such as polyreactivity, but accurate models require large, diverse, and high-quality labeled datasets, which are rarely publicly available and costly to produce. In this project, we develop a computational experimental design framework to simulate data acquisition. Sequences are represented using normalized Levenshtein distance to define similarity structure and embedded using ESM-2 (650M, 1280-dimensional) to capture biologically meaningful features. The goal is to identify the most informative protein sequences to label, thereby maximizing predictive performance while minimizing experimental cost.
BCE Team 2: Triple Helix:Machine Learning-Based Material Decomposition in Spectral Chest Radiography
Quentin Molina, Sujay Srinivasan, Braden White
Advisor: Dr. Lan Ma / Sponsor: Dr. Bahaa Ghammraoui
Chest computed tomography (CT) exams are among some of the most frequently performed, high-yield, diagnostic imaging exams globally. However, traditional scanning methods often struggle with efficiency and reliability in producing high-quality imaging results. Consequently, multiple acquisitions are necessary in delivering proper results, especially due to inept tissue differentiation by conventional CT scanning processes. To address these issues, our Capstone team designed a machine-learning based photon-counting computed tomography pipeline. Our pipeline delivers superior image resolution as well as enhanced material decomposition at a fraction of a traditional CT exam radiation dose.
Published May 15, 2026







