|
FOR IMMEDIATE RELEASE
July 26, 2006
 COLLEGE PARK, Md.—A substance found in crab shells is the key component in a nanoscale sensor system developed by researchers at the University of Maryland's A. James Clark School of Engineering. The sensor can detect minute quantities of explosives, bioagents, chemicals, and other dangerous materials in air and water, potentially leading to security and safety innovations for airports, hospitals, and other public locations. COLLEGE PARK, Md.—A substance found in crab shells is the key component in a nanoscale sensor system developed by researchers at the University of Maryland's A. James Clark School of Engineering. The sensor can detect minute quantities of explosives, bioagents, chemicals, and other dangerous materials in air and water, potentially leading to security and safety innovations for airports, hospitals, and other public locations.
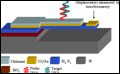
 Clark School engineers are using a substance called chitosan (pronounced "kite-o-san"), found in the shells of the Chesapeake Bay's famous blue crab, to coat components of the microscopic sensor system (see photo right). Clark School engineers are using a substance called chitosan (pronounced "kite-o-san"), found in the shells of the Chesapeake Bay's famous blue crab, to coat components of the microscopic sensor system (see photo right).
Crab lovers can hold on to their mallets—the crabs do not need to be harvested specifically for this purpose. The material is extracted from the crab shell waste.
Reza Ghodssi, associate professor in the Clark School's Department of Electrical and Computer Engineering and the Institute for Systems Research (ISR), and a member of the Maryland NanoCenter, is one of the investigators leading the project. He is joined by a multidisciplinary group: Gary Rubloff from ISR and the NanoCenter, Bill Bentley from the Fischell Department of Bioengineering and Greg Payne from the University of Maryland Biotechnology Institute (UMBI).
"Chitosan is interesting because it's a biological compound that can interact with a wide variety of substances, and also work well in a complex, sensitive device," Ghodssi says.
 Ghodssi's graduate students, Nathan Siwak, Stephan Koev, Jonathan McGee and Mike Fan, are helping to develop the nanoscale "system on a chip." It employs multiple miniature vibrating cantilevers, similar to diving boards (see photo above), which are coated with chitosan, plus optical sensing technology that can see when the cantilevers' vibrations change (such devices are called micro-electro-mechanical systems or MEMS). Ghodssi's graduate students, Nathan Siwak, Stephan Koev, Jonathan McGee and Mike Fan, are helping to develop the nanoscale "system on a chip." It employs multiple miniature vibrating cantilevers, similar to diving boards (see photo above), which are coated with chitosan, plus optical sensing technology that can see when the cantilevers' vibrations change (such devices are called micro-electro-mechanical systems or MEMS).
Different cantilevers can detect different substances and concentrations. When a targeted substance enters the device from the air or water, the chitosan on a specific cantilever interacts with the substance and causes that cantilever's vibration to change its characteristics. The optical sensing system sees the vibration change and indicates that the substance has been detected.
Ghodssi and his collaborators have recently submitted a proposal to the National Institutes of Health (NIH) to develop a sensor system to detect the presence of avian flu.
The technology was developed and initially tested at the Laboratory for Physical Sciences (LPS) in College Park, Md., and it is currently sponsored by LPS and the National Science Foundation (NSF).
"This is an exciting and complex microsystem that bridges biotechnology and nanotechnology to address critical needs of homeland security applications. My colleagues and I are expecting this work to become a product in the near future," says Ghodssi, who has to date filed for six patents on the technology.
Parts of this research were recently featured in the Journal of Micromechanics & Microengineering in April 2006 and the journal Biomacromolecules in November 2005.
NOTE TO EDITORS: Although they are microscopic in size, the devices can be seen in action through a microscope on a computer screen in the lab (see an example here).
More About Chitosan
Chitosan is derived from chitin, one of nature's most abundant biological compounds. Chitin makes up the shells of crabs and other crustaceans, insects, zooplankton and even the cell walls of mushrooms. It is both a polymer (a large molecule composed of repeating units) and produced by living things (biological). Chitin and its derivative chitosan are thus known as biopolymers. The researchers purchase chitosan in a purified, flake form from scientific supply companies.
Helpful Links
MEMS Sensors and Actuators Lab (MSAL) website: www.enee.umd.edu/MEMS
Prof. Reza Ghodssi's homepage:
www.ece.umd.edu/~ghodssi
Maryland NanoCenter:
http://www.nanocenter.umd.edu
University of Maryland Biotechnology Institute:
http://www.umbi.umd.edu
References
M. W. Pruessner, N. Siwak, K. Amarnath, S. Kanakaraju, W.-H. Chuang and R. Ghodssi, "End-coupled Optical waveguide MEMS Devices in the Indium Phosphide Material System," Journal of Micromechanics and Microengineering, Vol. 16, pp. 832-842, April 2006.
H. Yi, L-Q Wu, W. E. Bentley, R. Ghodssi, G. W. Rubloff, J. N. Culver, and G. F. Payne, "Biofabrication with Chitosan," Journal of Biomacromolecules, Vol. 6, No. 6, pp. 2881-2894, 2005.
About the A. James Clark School of Engineering
The Clark School of Engineering, situated on the rolling, 1,500-acre University of Maryland campus in College Park, Md., is one of the premier engineering schools in the U.S.
The Clark School's graduate programs are collectively the fastest rising in the nation. In U.S. News & World Report's annual rating of graduate programs, the school is 15th among public and private programs nationally, 9th among public programs nationally and first among public programs in the mid-Atlantic region. The School offers 13 graduate programs and 12 undergraduate programs, including two degree programs tailored for working professionals and one certification program.
The school is home to one of the most vibrant research programs in the country. With major emphasis in key areas such as communications and networking, nanotechnology, bioengineering, reliability engineering, project management, intelligent transportation systems and space robotics, as well as electronic packaging and smart small systems and materials, the Clark School is leading the way toward the next generations of advanced engineering technology.
Visit the Clark School homepage at www.eng.umd.edu.
About UMBI
With research centers in Baltimore, Rockville, and College Park, the University of Maryland Biotechnology Institute is the newest of 13 institutions forming the University System of Maryland. UMBI has 85 ladder-ranked faculty and a 2006 budget of $60 million. Celebrating the institution's 20th year of service to Maryland and the world, UMBI is led by microbiologist and former biotechnology executive Dr. Jennie C. Hunter-Cevera. For more information visit www.umbi.umd.edu.
####
|
 |

|



 COLLEGE PARK, Md.—A substance found in crab shells is the key component in a nanoscale sensor system developed by researchers at the University of Maryland's A. James Clark School of Engineering. The sensor can detect minute quantities of explosives, bioagents, chemicals, and other dangerous materials in air and water, potentially leading to security and safety innovations for airports, hospitals, and other public locations.
COLLEGE PARK, Md.—A substance found in crab shells is the key component in a nanoscale sensor system developed by researchers at the University of Maryland's A. James Clark School of Engineering. The sensor can detect minute quantities of explosives, bioagents, chemicals, and other dangerous materials in air and water, potentially leading to security and safety innovations for airports, hospitals, and other public locations. Clark School engineers are using a substance called chitosan (pronounced "kite-o-san"), found in the shells of the Chesapeake Bay's famous blue crab, to coat components of the microscopic sensor system (see photo right).
Clark School engineers are using a substance called chitosan (pronounced "kite-o-san"), found in the shells of the Chesapeake Bay's famous blue crab, to coat components of the microscopic sensor system (see photo right). Ghodssi's graduate students, Nathan Siwak, Stephan Koev, Jonathan McGee and Mike Fan, are helping to develop the nanoscale "system on a chip." It employs multiple miniature vibrating cantilevers, similar to diving boards (see photo above), which are coated with chitosan, plus optical sensing technology that can see when the cantilevers' vibrations change (such devices are called micro-electro-mechanical systems or MEMS).
Ghodssi's graduate students, Nathan Siwak, Stephan Koev, Jonathan McGee and Mike Fan, are helping to develop the nanoscale "system on a chip." It employs multiple miniature vibrating cantilevers, similar to diving boards (see photo above), which are coated with chitosan, plus optical sensing technology that can see when the cantilevers' vibrations change (such devices are called micro-electro-mechanical systems or MEMS).